Muireann Duffy
As rising Covid-19 cases have plunged European countries back into lockdown, a 'light at the end of the tunnel' has become a common theme in the announcement of increased restrictions.
Last night UK prime minister Boris Johnson became the latest leader to evoke the sentiment, announcing a third national lockdown for England expected to last until mid-February.
On December 30th, when Taoiseach Micheál Martin announced the return to a full-scale Level 5 lockdown for the country, he said: "The next month, as we face into these strictest controls in the depth of winter, is going to be very tough for everyone.
"But there is another key difference between then and now. After a year of the most extraordinary effort by the most talented scientific minds on earth, we have safe and effective vaccines."
Although public health experts have warned it will take some time for the vaccination processes to positively impact daily case numbers, news of their development, approval and administration has offered hope amid almost constant gloom.
As a member state, Ireland will gain access to vaccines the EU has struck deals with manufacturers for, following their approval by the European Medicines Agency (EMA).
Currently, the EU has six Advance Purchase Agreements (APAs), which entitles Ireland to approximately 1.11 per cent or 14.4 million doses of the EU's total stock. This figure was determined on a pro-rata basis, based on Ireland's population.
The below information was included in the Department of Health's 'National Covid-19 Vaccination Programme: Implementation Plan' published on December 15th:
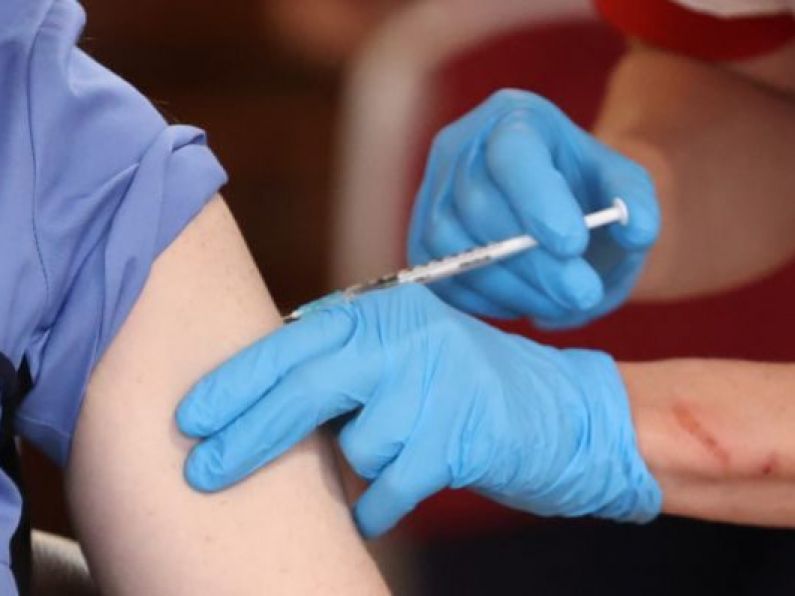
At the end of last year, the Pfizer/BioNTech vaccine became the first of the six candidates to gain approval from the EMA.
The first doses of the vaccine arrived in EU member states on St Stephen's Day, with the first shot being administered in the Republic on December 29th.
Next in line is the Moderna vaccine, which is likely to be the second candidate to get the green light for distribution in the EU.
The EMA brought forward their scheduled meeting by two days to discuss the Moderna vaccine on Monday and their approval is expected in the coming days.
Like the Pfizer/BioNTech jab, the Moderna vaccine is administered in two doses, but the later can be more easily distributed as it does not need to be stored at ultra-low temperatures.
According to the EMA website, the evaluation of the conditional marketing authorisation for Moderna vaccine began on December 1st.
Additional data
The Moderna vaccine has already been approved for emergency use in the UK, alongside another of the EU's candidates, the AstraZeneca vaccine developed with Oxford University.
The AstraZeneca jab is being considered by the EMA, but the process has been delayed as the regulatory body said it requires additional information, with deputy executive director Noel Wathion saying they may not make a decision until some time in February.
If the AstraZeneca vaccine gets the go-ahead, it will be a major milestone in the vaccination process as it will be the first shot which can be stored in a fridge, making it even easier to distribute.
Rolling reviews of both the AstraZeneca and Jassen Pharmaceuticals (J&J) shots, the only one of the six candidates which requires just one dose, are being carried out by the regulator, having begun on October 1st and December 1st respectively.
The EMA site also shows the CureVac shot received advice from the regulator during its non-clinical phase of development, but has not yet been listed as a vaccine being considered by the EMA's Committee for Medicinal Products for Human Use (CHMP).
According to the European Commission, the Sanofi/GSK vaccine was undergoing Phase 1/2 trials in September, with Phase 3 trials due to commence before the end of last year.
Following that timetable, the companies are aiming for their jab to be ready for distribution in the EU, pending EMA approval, by the second half of this year.
Dosage schedule
Yesterday, Professor Karina Butler confirmed Ireland would not be following the UK in delaying the second dose of the vaccine.
The vaccines are recommended to be administered approximately 21 days apart, with the first dose supplying a lower level of protection against the virus, before the second dose increases the efficacy.
The UK has chosen to give a larger number of people their first dose of the vaccine in the hope that a higher number of people being partially protected will have a greater impact on transmission than a smaller number of people having higher protection.
The change in tack from the NHS has been widely questioned and the impact of delaying second doses has since been examined by the National Immunisation Advisory Committee (NIAC).
Speaking at yesterday's Covid-19 briefing, chair of the Committee, Professor Karina Butler said Ireland would not be adopting the UK's altered approach as the first in line to receive the vaccine ought to receive the highest protection possible, including healthcare workers and over 65s living in long-term care facilities.
"They are the group you don't want to compromise the efficacy of the vaccine [with]," Prof Butler said, adding: "We did not find evidence to alter the current schedule."